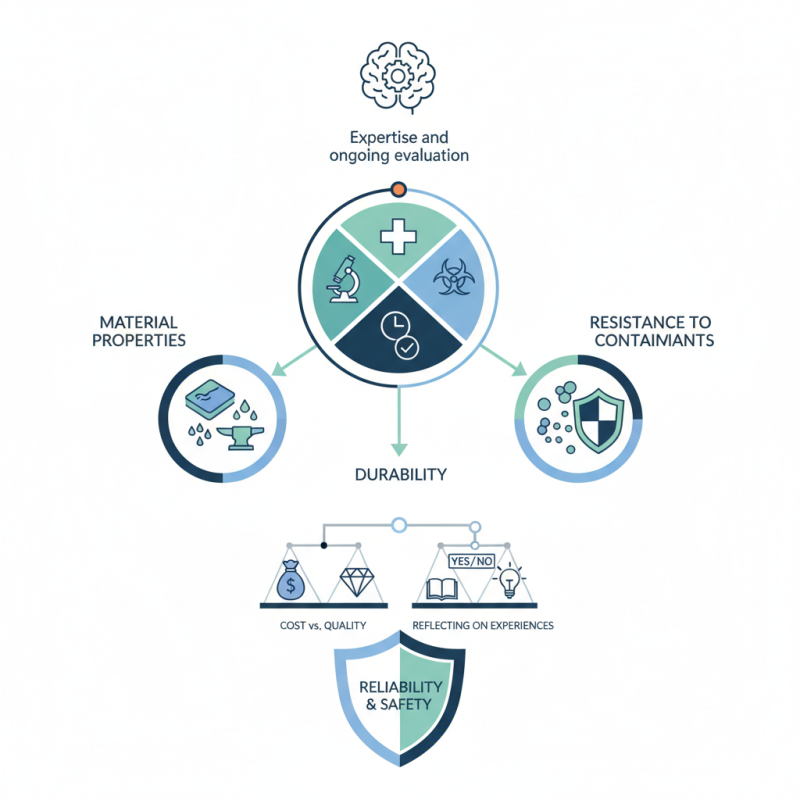
Selecting materials for sterile environments is a critical process that demands careful consideration. Professionals in healthcare and laboratory settings understand the importance of maintaining sterile conditions. Effective material selection can significantly influence infection control and overall safety.
In this context, knowing how to select materials for sterile environments becomes essential. Factors such as material properties, durability, and resistance to contaminants play a vital role in this decision-making process. For example, certain plastics are preferred over metals due to their non-porous nature and ease of sterilization.
However, challenges in this selection process can arise. The balance between cost and quality often leads to difficult choices. Not all materials perform equally in every situation. Reflecting on previous experiences can help improve future decisions. Thus, understanding how to select materials for sterile environments is not just a technical task; it requires expertise and ongoing evaluation to ensure reliability and safety.
Material selection in sterile environments is crucial to preventing contamination. Studies show that over 60% of surgical site infections are linked to improper material choices. Selecting the right materials helps maintain sterility throughout medical procedures. For instance, using non-porous surfaces can reduce bacterial proliferation.
The importance of biocompatibility is another key factor. Reports indicate that materials with low toxicity rates significantly lower infection risks. For example, materials rated Class VI in the medical category are preferred. However, determining the lifecycle and durability of these materials often poses challenges. Many facilities overlook this, potentially compromising effectiveness.
Quality control is essential in sterile environments. Data reveals that nearly 30% of recalled medical devices were due to material failures. Regular audits and thorough testing can mitigate these risks. Facilities should focus on refining their selection processes to ensure long-term reliability. Selecting materials is not just about compliance; it requires a deep understanding of both function and safety.
Selecting materials for sterile environments is critical to ensure safety and efficacy. The key characteristics required for sterile materials include biocompatibility, non-reactivity, and ease of sterilization. Biocompatibility guarantees that materials do not produce adverse reactions. Non-reactivity ensures that materials do not interact negatively with other substances. Easy sterilization processes reduce the risk of contamination.
Tips: Always evaluate the material's resistance to environmental factors. High humidity can compromise some materials. Regular testing for integrity is crucial. Contamination can happen even with minor defects.
When considering material selection, focus on durability. Sterile materials must withstand frequent handling and cleaning. Monitor performance over time; some materials degrade quicker than expected. Be aware of changes in appearance or function. Regular assessments can uncover hidden weaknesses. Remember that no single material is perfect, and it’s essential to be open to adjustments.
Tips: Document each material's performance. This creates a reliable history for future reference. Transparency in this process builds trust and ensures ongoing compliance.
| Material Type | Biocompatibility | Sterilization Compatibility | Chemical Resistance | Durability | Cost |
|---|---|---|---|---|---|
| Polypropylene | Good | Yes | Moderate | High | Low |
| Medical Grade Silicone | Excellent | Yes | High | Moderate | High |
| Stainless Steel | Good | Yes | Excellent | Very High | Moderate |
| Polyethylene | Fair | Yes | Low | Moderate | Very Low |
| Glass | Excellent | Yes | High | Very High | Moderate |
Selecting the right materials for sterile environments is crucial. Different settings require specific types of materials to meet sterility standards. Commonly used materials include stainless steel, glass, and specialized plastics. Each material has unique properties that can impact cleanliness and safety.
Stainless steel is a popular choice due to its durability and resistance to corrosion. A study from the American Journal of Infection Control reported that stainless steel surfaces harbor fewer bacteria compared to other materials. However, it can be expensive and may require intensive cleaning protocols.
Glass is non-porous and can be sterilized effectively, but its fragility poses challenges in some environments. On the other hand, specialized plastics offer flexibility and lightweight options, but their potential for surface degradation raises concerns in long-term usage.
Evaluating these materials should be done critically. While stainless steel excels in durability, it's crucial to consider maintenance costs. Glass might be less practical due to its breakability, despite its sterilization benefits. Specialized plastics can break down over time, leading to compromised sterility. Assessing how well each material fits the environment can help make informed decisions. Data from the Health Facilities Management journal indicates that improper material selection can lead to increased infection rates, emphasizing the need for careful evaluation.
Selecting materials for sterile environments is crucial. Environmental factors can greatly influence material choice. Assessing conditions like humidity, temperature, and exposure to chemicals is essential. For example, high humidity may cause some materials to degrade faster.
Temperature fluctuations can impact material integrity as well. Certain substances may expand or contract, leading to potential failures. It’s vital to test materials under varying conditions to ensure reliability. Additionally, exposure to certain chemicals can compromise material performance.
Relying solely on generic data may not suffice. Evaluating materials in real-world scenarios is often necessary. Some materials might perform well in controlled tests but fail under actual operational stresses. Regular reviews of performance data can help identify these discrepancies. Maintaining a proactive approach ensures materials remain suitable over time.
In sterile environments, selecting appropriate materials is crucial. Effective material testing and validation help ensure sterility. Various factors need evaluation including biocompatibility, chemical resistance, and durability. Testing should begin with the selection of relevant standards and guidelines. This mitigates risks associated with material failures.
A robust validation process involves thorough microbiological testing. Using a variety of test methods enhances reliability. For example, consider using sterility testing, endotoxin testing, and bioburden assessments. Each method provides unique insights into material performance. Yet, even with rigorous testing, unexpected results can occur. It’s essential to analyze these failures critically. Reflection on why certain materials failed can guide better choices in the future.
Collaboration among teams is vital during testing and validation. Engage scientists, engineers, and quality control experts. Their diverse expertise can enhance the testing approach. Communication is key to understanding potential weaknesses in material choices. This, in turn, helps in reinforcing standards. Continuous improvement is necessary in adopting best practices to ensure that sterile environments remain effective and reliable.
: Material selection is crucial to prevent contamination and reduce surgical site infections.
The key characteristics include biocompatibility, non-reactivity, and ease of sterilization.
High humidity can compromise certain materials and accelerate degradation.
Quality control helps identify material failures, which are linked to many recalled medical devices.
Regular testing is crucial, as minor defects can lead to contamination.
Factors like temperature and chemical exposure can significantly influence material integrity and performance.
Yes, materials that perform well in controlled tests may fail during real-world use.
Yes, documenting performance creates a reliable history for future reference.
Facilities should regularly assess materials and be open to adjustments.
Many facilities overlook lifecycle assessment, which can compromise material effectiveness.
When considering how to select materials for sterile environments, it is crucial to understand the significance of material selection in maintaining sterility and preventing contamination. Key characteristics such as biocompatibility, resistance to temperature fluctuations, and ease of sterilization must be identified to ensure the materials meet the stringent requirements of sterile settings.
Furthermore, an evaluation of commonly used materials, along with an assessment of environmental factors influencing material choice, plays a vital role in this process. Best practices in material testing and validation are also essential to guarantee that the selected materials consistently uphold the required sterility standards. By integrating these elements, one can effectively ensure the integrity of sterile environments.

 Ultrivex Medical
Ultrivex Medical