














| Still deciding? Get samples of $ ! US$ 0/Piece Request Sample |
U-PACK Interventional Surgical Kit is a cost-effective, sterile-packed solution designed for minimally invasive radiological procedures. This kit contains 25 essential components compliant with hospital standards, including radiation-protective drapes, specialized instruments, and absorbent textiles. All items are pre-sterilized using EO gas and individually packed for single-use safety.

| Parameter | Specification |
|---|---|
| Product Name | Interventional Surgical Kit (Ver.02) |
| Sterilization | EO Gas, Valid 3 years |
| Material | SMS Nonwoven (45g), Composite Membrane (70g) |
| Gown Size | XL (3 pcs) |
| Key Components | 5-Hole Drape, Radiation Covers, Syringes, Basins |
| Drape Hole Size | Ø10cm (Small) / Multi-hole (Large) |
| Packaging | 1 kit/sterile bag → 20 kits/carton |
| Production Standard | ISO 13485, GB/T 19633.1 |
| No. | Component | Specification | Qty | Details |
|---|---|---|---|---|
| 1 | Waste Bag | 80×100cm | 1 | PE material |
| 2 | Surgical Gown | XL | 3 | 45g SMS nonwoven, reinforced |
| 3 | Baffle Cover | 75×90cm | 1 | Green PE bag packed |
| 4 | Machine Head Cover | 100×100cm | 1 | Green PE bag packed |
| 5 | Wire Guide Basin | 4500ml | 1 | Green color |
| 6 | Medicine Bowls | 500ml | 3 | Green/Yellow/White |
| 7-9 | Syringes | 10ml/20ml | 3 | Luer-lock without needle |
| 10 | Gauze Sponges | 10×8cm-8ply | 25 | High absorbency |
| 11 | Sponge Brushes | 50×50×25mm | 4 | Yellow sponge |
| 13 | Hand Towels | 56×75cm | 3 | Hydroentangled fabric |
| 14 | Fenestration Drapes | 50×60cm | 2 | Ø10cm hole, composite membrane |
| 16 | Multi-Hole Drape | 200×375cm | 1 | 5 holes, adhesive film backing |
| 19 | Wrapping Cloth | 150×240cm | 2 | 70g semi-composite |



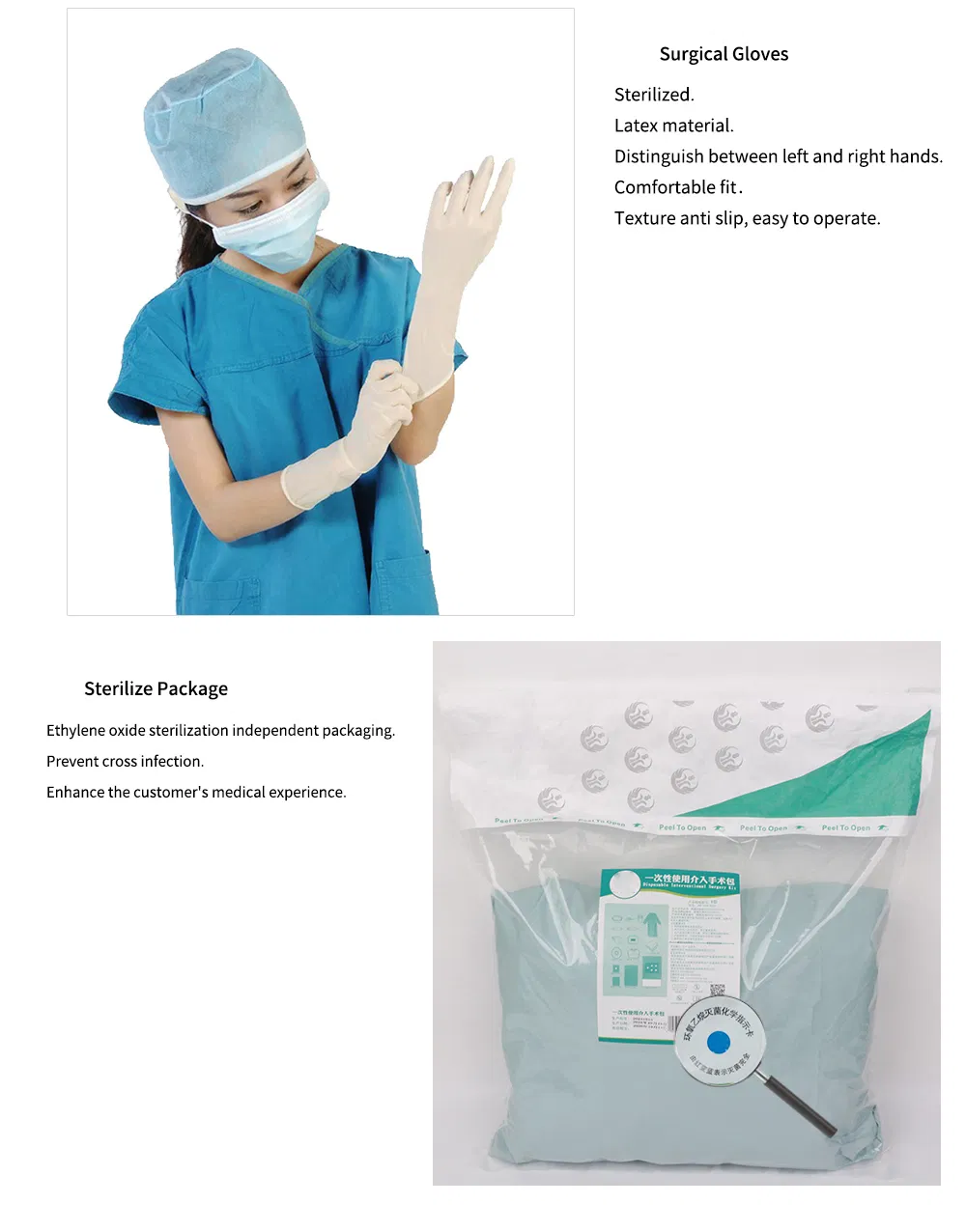


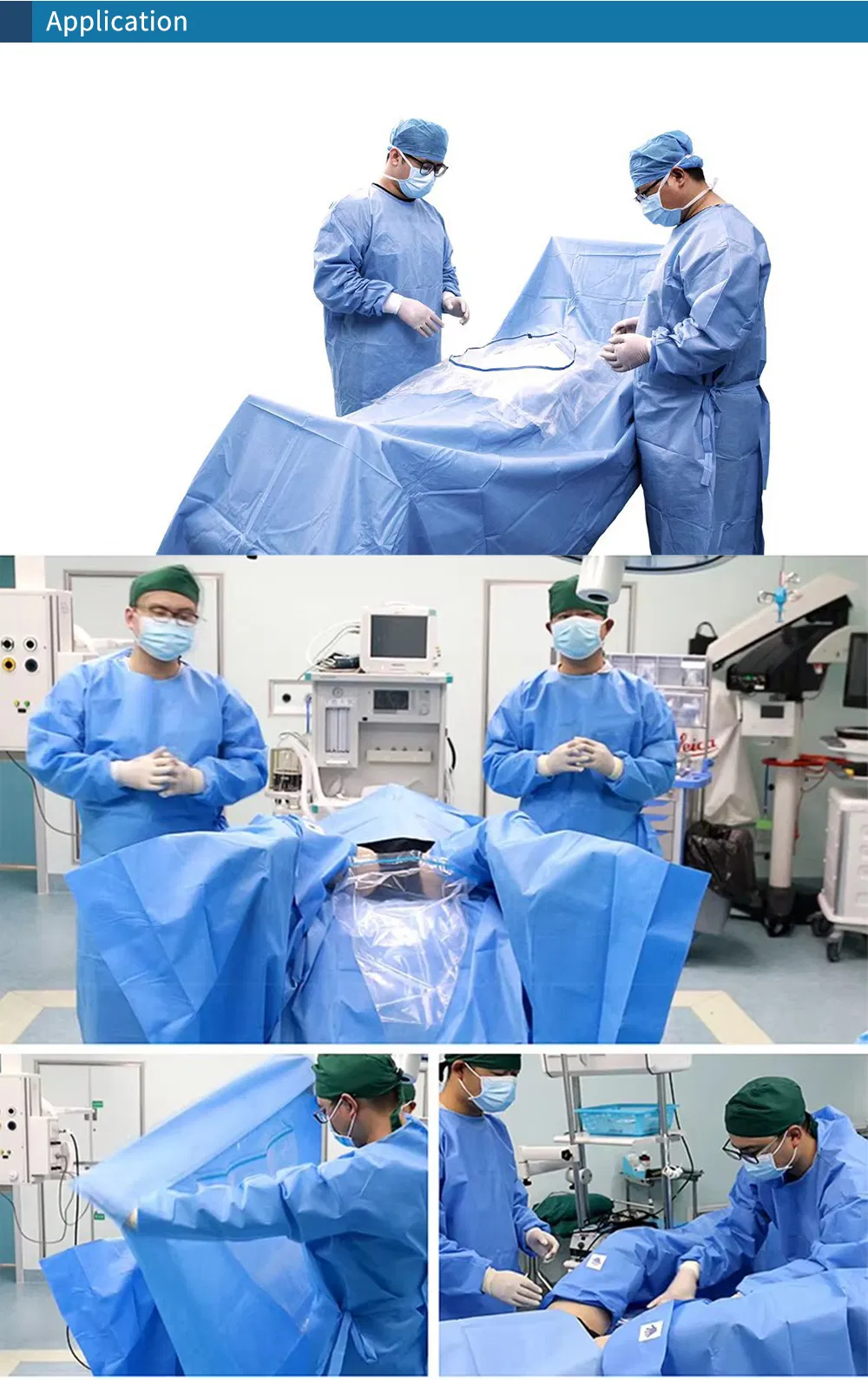



Our Surgical Packs are specially designed to combine key standard components for specific procedures within surgical and related specialties. These packs contain multiple individual disposables, fully sterilized and ready for immediate use. By using standard surgical packs, medical facilities can ensure a safe, consistent standard of care while promoting process efficiency compared to managing items separately. All packs are manufactured following strict ISO and CE standards.
 Ultrivex Medical
Ultrivex Medical